The Didomi Behavioral Model
A Systems Biology Framework for Neurobehavioral Dynamics and Allostatic Regulation
Abdul Martinez
Didomi Research
didomiresearch.org
Working Paper — February 2026
Version 1.2
Official HTML version of the Didomi Behavioral Model (Version 1.2, February 2026).
For citation and archival purposes, the PDF version is the version of record.
Abstract
The prediction and management of human behavior have traditionally relied on psychological constructs that treat behavior as a flexible output, shaped primarily by cognition, learning history, and environmental contingencies. In these frameworks, the body is often treated as a passive backdrop. The Didomi Behavioral Model (DBM) reframes this assumption by presenting behavior as an emergent property of five evolutionarily conserved, homeostatic regulatory systems: Energy Management, Recovery, Status, Attention, and Connection.
Drawing on systems biology, mitochondrial bioenergetics, neuroendocrine allostasis, and large-scale functional network dynamics, the DBM offers a mechanistic account of how physiological constraints shape “volitional” behavior. The model details how chronic stress and energy deficits initiate cascades of dysregulation, including glucocorticoid resistance, neuroinflammation, striatal dopamine suppression, and salience network rigidity, and argues that many so-called “maladaptive” behaviors are, in fact, functional biological solutions to unsustainable internal demands.
We outline the neurobiological substrates of each system, describe the feedback loops that couple them into higher-order dynamics (e.g., burnout and social defeat cascades), and propose a clinical shift from behavioral modification toward capacity restoration and accommodation. Finally, we position DBM in relation to existing frameworks such as allostatic load, Polyvagal Theory, Predictive Processing, the Free Energy Principle, and social rank models, and identify testable predictions and research directions.
Keywords: Systems Biology, Allostasis, Mitochondrial Bioenergetics, Neuroinflammation, Salience Network, Oxytocin–Dopamine Interactions, Social Defeat, Behavioral Ecology.
1. Introduction: The Biological Basis of Volition
1.1 Behavior as Constrained Output
Most clinical and applied behavioral frameworks, ranging from operant conditioning to cognitive–behavioral approaches, implicitly treat behavior as a malleable output of cognition and contingencies. Interventions are designed as if the primary levers of change are beliefs, thoughts, reinforcement schedules, and skills training. These models have generated powerful tools and substantial evidence; however, they are implicitly biologically permissive: they assume that the nervous system has enough energy, flexibility, and structural integrity to implement new behavioral patterns once the “right” cognitive or environmental inputs are applied.
The DBM challenges this assumption. It asserts that behavior is strictly bounded by the real-time functional capacity of underlying neurobiological systems. When energy is depleted, inflammation is high, social threat is chronic, or large-scale brain networks lose flexibility, the space of possible behaviors narrows dramatically. “Non-compliance,” “lack of motivation,” or “poor self-control” may reflect a state of constrained biological capacity, not a deficit of will or insight.
In this view, behavioral interventions that ignore physiological capacity function like asking a machine to produce more output after it has already entered thermal shutdown. This mismatch between demands and capacity contributes to treatment non-response, relapse, and the chronicity of many neurobehavioral conditions.
1.2 Evolutionary Prioritization and the Logic of Survival
The DBM is grounded in evolutionary neuroscience and behavioral ecology. It assumes that the brain’s regulatory architecture was optimized not for happiness or flourishing but for survival and reproduction in uncertain environments.
Under acute threat or resource scarcity, the organism shifts resources away from metabolically costly, high-level functions (long-term planning, curiosity, exploration, nuanced social engagement) toward low-cost, high-survival-payoff strategies (vigilance, withdrawal, defensive aggression, energy conservation). From a systems perspective, this is not a pathological error; it is the expected behavior of a system evolved to minimize catastrophic loss.
The DBM thus interprets symptoms such as fatigue, anhedonia, distractibility, irritability, and emotional numbing as predictable outputs of systems operating under high allostatic load. These behaviors are the “best available solutions” under constraints, not arbitrary deviations from a norm.
In this context, allostasis refers to the process by which the organism maintains stability through change, while allostatic load denotes the cumulative physiological cost incurred when adaptive systems are repeatedly or chronically engaged beyond their recovery capacity.
1.3 Systems Biology and Nonlinear Neurobehavioral Dynamics
Biological regulation is inherently nonlinear. Small perturbations can produce large downstream effects when systems are near critical thresholds; chronic low-level stressors can accumulate into tipping points via positive feedback loops. Systems biology and System Dynamics modeling offer tools for conceptualizing such complexity, emphasizing:
- Feedback loops (reinforcing vs. balancing),
- Delays and lags between input and observable output,
- Emergent properties not predictable from individual components,
- Sensitivity to initial conditions and system structure.
The DBM adopts this lens, treating behavior as the emergent expression of interacting regulatory systems rather than the direct product of isolated variables. Instead of asking “Which single factor caused this behavior?”, DBM asks, “Which feedback structure is currently dominating the system?”
1.4 From Psychological to Neurobiological Models of Control
Previous integrative frameworks, such as the allostatic load model, Polyvagal Theory, and various neurocognitive control models, have emphasized specific subsystems (stress hormones, autonomic hierarchy, executive control, or prediction errors). The DBM aims to organize these insights into a unified, tractable framework that remains close to biology yet is usable in clinical and applied settings.
To do this, the DBM reduces the complexity of human regulation into five deeply coupled systems: Energy Management, Recovery, Status, Attention, and Connection. Each is anchored in specific organ systems, neural circuits, and signaling molecules, yet each also manifests as recognizable behavioral patterns and subjective experiences. Understanding these five systems, and how they interact, allows a large fraction of human behavior, especially under stress, to be mapped to biological constraints rather than moral or character judgments.
2. The Didomi Behavioral Model
2.1 Overview of the Five Regulatory Systems
The DBM proposes that human behavior emerges from the interaction of five homeostatic systems:
- Energy Management System (EMS) – Regulates metabolic resources and arousal levels, largely through mitochondrial function and neurometabolic coupling.
- Recovery System (RS) – Coordinates stress responses and restoration via the HPA axis, autonomic balance, and immune/inflammatory processes.
- Status System (SS) – Monitors social rank and relative value, shaping behavioral strategy through serotonergic and dopaminergic mechanisms.
- Attention System (AS) – Filters sensory input and assigns priority through large-scale brain networks, especially the salience, default mode, and executive networks.
- Connection System (CS) – Regulates attachment, trust, and social proximity through oxytocin–dopamine interactions and related social neuropeptide systems.
Each system has a core biological function, a set of canonical failure modes, and characteristic behavioral signatures. While analytically separable, they are tightly coupled in practice.
Within the DBM, the Energy Management System functions as a central energetic gatekeeper, mediating whether evaluative signals from Attention, Status, Connection, and Recovery systems are translated into action. These systems assess salience, social consequence, relational reward, and physiological threat, respectively; the EMS determines whether sufficient metabolic resources are available to support action without jeopardizing survival. Behavior emerges only when both evaluative signals and energetic feasibility converge.
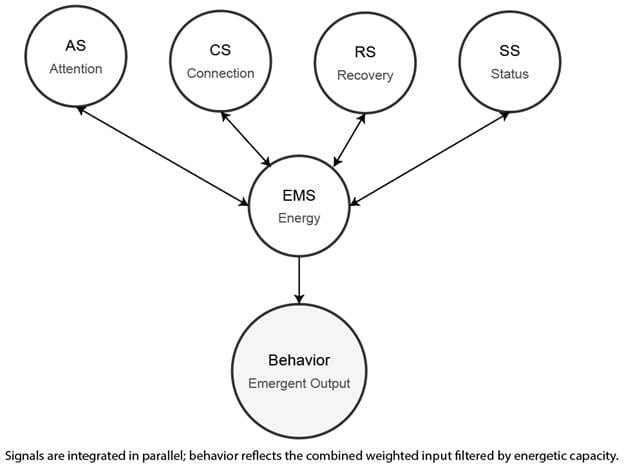
[Figure 1: Didomi Behavioral Model — In the DBM, the Energy Management System integrates simultaneous evaluative signals from attention, status, connection, and recovery systems. These signals are weighted dynamically and assessed against current metabolic capacity. Behavior is released only when the combined signal exceeds an energetic feasibility threshold, making behavioral output an emergent property of parallel system integration rather than a linear decision process.]
2.1.1 Ontological Commitments
The DBM makes a strict distinction between four categories of phenomena:
- Resources: Energy is a real, finite biological resource. Energy provisioning by the EMS is the only process that enables action. No energy, no action—regardless of signals, values, or rewards.
- Signals: Reward signals, wanting, threat salience, social value cues, attachment signals, and interoceptive states (fatigue, hunger, pain) carry information. They do not cause behavior. Signals are informational, not causal.
- Evaluation: Each of the five regulatory systems evaluates inputs in its domain—feasibility (EMS), threat and recovery (RS), social value and rank (SS), salience and priority (AS), and safety and attachment (CS). Evaluation biases and annotates potential action but does not initiate it.
- Action: Action occurs only when the EMS provisions energy following evaluation. All other processes prepare, bias, or annotate action.
This hierarchy has a critical implication: when the paper describes a neurotransmitter, hormone, or inflammatory signal as “promoting,” “biasing,” or “shifting” behavior, this should be read as signal-level influence subject to evaluative gating—not as direct causation.
2.1.2 The Status of Reward in DBM
DBM does not include a reward system as a distinct regulatory component. This is a deliberate architectural decision, not an oversight.
What DBM rejects is the treatment of reward as a goal, a driver of behavior, a privileged system, or a motivational controller. What DBM retains is that reward signals exist as informational readouts: they annotate that energy was provisioned and an action was completed. Reward signals contribute to learning and future evaluation but are post-decisional and non-causal—they do not initiate action, override feasibility constraints, or function as goals.
This commitment distinguishes DBM from reinforcement learning models and reward-maximization frameworks, in which reward signals play a privileged causal role. In DBM, the question is not “What does the organism want?” but “What can the organism afford?”
2.2 Relationship to Existing Frameworks
The DBM is not intended to supplant existing models but to organize them into a systems-based scaffold:
- Allostatic load concepts are primarily reflected in the EMS and RS.
- Polyvagal Theory maps closely onto aspects of the RS and CS.
- Social rank and social defeat literature are formalized within the SS.
- Predictive Processing and Triple Network models are operationalized in the AS.
- Social Baseline Theory and attachment models align with the CS (Beckes & Coan, 2011).
The DBM’s contribution is to place these strands within a single, feedback-oriented architecture, making explicit how changes in one domain (e.g., mitochondrial exhaustion) propagate across stress systems, social rank biology, and network dynamics to produce recognizable behavioral phenotypes.
3. The Five Regulatory Systems in Detail
3.1 The Energy Management System (EMS)
Core Function: The EMS allocates metabolic resources to support neural computation and bodily function. It determines how much “human operating system” can be online at any given time.
In addition to regulating metabolic capacity, the EMS mediates whether evaluative signals from other systems can be translated into action.
3.1.1 Mitochondria as Sensors and Decision Nodes
Mitochondria are not mere “power plants”; they integrate signals about nutrient availability, oxidative stress, toxins, and hormonal states. Changes in mitochondrial function alter:
- ATP availability for high-cost processes (PFC activity, sustained attention, complex motor patterns),
- Reactive oxygen species (ROS) signaling,
- Retrograde signaling to the nucleus, shifting gene expression from growth and plasticity to defense and conservation phenotypes.
These mechanisms provide a cellular-level explanation for phenomena often labeled “psychological,” such as chronic fatigue, loss of motivation, and cognitive fog.
3.1.2 Sleep, Prefrontal Control, and Energetic Trade-Offs
Sleep deprivation reliably reduces metabolic activity in the prefrontal cortex and disrupts its connectivity with limbic structures. The result is:
- Impaired top-down regulation of the amygdala,
- Increased reactivity to emotional stimuli,
- Reduced capacity for delayed gratification and complex decision-making.
From a DBM perspective, a chronically depleted EMS forces the system to shed high-cost functions first, executive control, long-term planning, nuanced empathy, well before more primitive circuits fail.
3.1.3 Behavioral Manifestations of EMS Dysregulation
EMS underload (depletion) is experienced as:
- Fatigue, “tired but wired”,
- Reduced tolerance for complexity and ambiguity,
- Preference for simple, low-effort tasks and immediate rewards.
Attempts to impose high-demand behavioral change in this state often provoke shutdown, impulsivity, or emotional volatility—not because of poor motivation but due to energetic constraints.
3.2 The Recovery System (RS)
Core Function: The RS coordinates the organism’s response to stressors and its return to baseline, integrating HPA axis activity, autonomic state, and immune/inflammatory processes.
3.2.1 HPA Axis Dynamics and Recovery Lag
Under stress, the hypothalamus–pituitary–adrenal (HPA) axis mobilizes cortisol and other mediators to support short-term adaptation. With chronic activation, structural and functional changes occur (e.g., adrenal hypertrophy, altered feedback sensitivity), producing a recovery lag: physiology remains in a “stressed” configuration long after external demands end.
This lag means that individuals can appear “overreactive” to minor triggers when, in fact, their baseline has shifted upward in arousal and inflammatory tone.
3.2.2 Glucocorticoid Resistance and Neuroinflammation
Chronic cortisol exposure leads immune cells to downregulate glucocorticoid receptors, blunting negative feedback and permitting a pro-inflammatory state characterized by elevated cytokines such as IL-6 and TNF-α. These mediators can:
- Cross the blood–brain barrier,
- Alter neurotransmitter metabolism (notably dopamine and serotonin),
- Induce “sickness behavior”: social withdrawal, anhedonia, psychomotor slowing, and energy conservation.
Thus, RS dysregulation directly feeds into Status and Connection systems via inflammation-mediated suppression of reward circuitry.
3.2.3 Autonomic Hierarchy and Its Ongoing Debates
The DBM incorporates the autonomic hierarchy described in Polyvagal Theory, which organizes autonomic responses into three broad modes: (1) ventral-vagal social engagement, (2) sympathetic mobilization, and (3) dorsal-vagal metabolic conservation.
While this framework has been influential for conceptualizing adaptive and defensive states, it is also the subject of ongoing empirical debate, particularly regarding the universality of its phylogenetic claims across species, the specificity of vagal pathways involved, and variability across individual nervous systems and environmental conditions.
Our use of Polyvagal principles is therefore functional rather than taxonomic: the DBM focuses on observable patterns of autonomic state transitions and their behavioral consequences, without assuming full resolution of the theoretical debates in the literature.
Notably, aspects of Polyvagal Theory, particularly claims regarding respiratory sinus arrhythmia (RSA) as a direct index of vagal tone, have been questioned (e.g., Grossman & Taylor, 2007), underscoring the importance of interpreting autonomic markers cautiously and contextually.
3.3 The Status System (SS)
Core Function: The SS estimates social rank, relative value, and “place in the group,” shaping behavioral strategy: whether to compete, cooperate, defer, or withdraw.
3.3.1 Social Defeat Stress and Structural Remodeling
Animal and human studies of social defeat show consistent neural changes:
- Dendritic atrophy in the medial PFC and hippocampus,
- Dendritic hypertrophy in the amygdala and other fear-related regions.
This pattern favors threat detection and avoidance over flexible planning and exploration, a logical adaptation when repeated social encounters result in defeat or humiliation.
3.3.2 Serotonin, Rank, and Bioenergetic Modulation
While serotonin (5-HT) is widely recognized for its role in mood regulation, it also participates in metabolic regulation through pathways such as SIRT1–PGC-1α, which influence mitochondrial biogenesis and neuronal energy allocation. Evidence from social dominance and social defeat paradigms suggests that 5-HT modulates behavioral strategy (e.g., risk-taking, impulsivity, deference), but this modulation is multifaceted and context-dependent, shaped by genetic background, developmental history, and environmental context.
In the DBM, we interpret serotonergic shifts not as fixed “bioenergetic instructions,” but as one regulatory channel through which perceived status can influence metabolic prioritization—an influence that interacts with other hormonal, environmental, and social cues rather than determining them unilaterally.
3.3.3 Dopamine and Rank-Dependent Strategy
Social rank is associated with striatal D2/3 receptor availability and dopaminergic tone. High status is linked to:
- Greater willingness to incur risk for potential gain,
- Higher exploratory behavior,
- Greater sense of agency.
Low status or status loss reduces dopaminergic tone, biasing the individual toward submissive, risk-averse, and withdrawal-oriented strategies. These patterns can be misread as “lack of ambition” or “poor motivation” when they are in fact economical strategies under perceived social threat.
3.4 The Attention System (AS)
Core Function: The AS filters incoming information and allocates cognitive resources by orchestrating large-scale brain networks.
3.4.1 Triple Network Model
The DBM adopts the Triple Network Model (Menon, 2011) as its primary architecture for the AS:
- Default Mode Network (DMN): Self-referential and internally focused processes.
- Central Executive Network (CEN): Goal-directed cognition, working memory, and problem solving.
- Salience Network (SN): Detection of relevant stimuli and switching between DMN and CEN.
Healthy functioning requires fluid switching between these networks based on demands.
3.4.2 Salience Rigidity and Threat Bias
Under chronic stress, anxiety, or burnout, the SN (anchored in the anterior insula and dorsal ACC) becomes hyperactive and less flexible. Consequences include:
- Over-assignment of threat salience to neutral cues,
- Difficulty disengaging from perceived dangers,
- Impaired switching from internal rumination (DMN) to task focus (CEN).
This produces the familiar state of being “tired and wired”: exhausted yet unable to disengage, prone to distraction, and overwhelmed by minor demands.
3.4.3 Cortisol, Connectivity, and Rumination
Acute and chronic stress modify functional connectivity patterns, often increasing SN–DMN coupling. This configuration biases the brain toward internal, self-referential processing, such as worry and rumination, at the expense of externally focused task engagement. In DBM terms, the AS becomes locked into a high-cost, low-yield configuration, increasing the metabolic burden on the EMS.
3.5 The Connection System (CS)
Core Function: The CS manages attachment, trust, and proximity to others. It determines whether the organism perceives “being with others” as safe and rewarding or dangerous and depleting.
3.5.1 Oxytocin–Dopamine Heteromers and Social Reward
A core mechanism in the DBM is the formation of heteroreceptor complexes between oxytocin receptors (OTR) and dopamine D2 receptors in the nucleus accumbens and related structures (Guidolin et al., 2025). In these complexes:
- Oxytocin binding alters D2 receptor conformation,
- This allosterically increases D2 receptor affinity for dopamine,
- The result is an amplified reward signal for social interactions that are perceived as safe and affiliative.
This mechanism transforms social connection from a psychological metaphor into a biochemically privileged source of reward.
3.5.2 Isolation, Reward Blunting, and Motivational Collapse
When social connection is absent, threatening, or chronically unreliable, the CS cannot effectively recruit this oxytocin–dopamine synergy. Consequences include:
- Blunted reward from everyday activities,
- Increased reliance on non-social rewards (substances, compulsive behaviors, digital stimulation),
- Vulnerability to depressive anhedonia and burnout.
From a DBM perspective, reconnection is not merely “supportive”; it functions as a pharmacological-grade intervention on the reward circuitry.
3.6 Cross-System Integration: High-Capacity States and Positive Adaptation
While much of the DBM emphasizes dysregulation under chronic stress or resource scarcity, the same five systems also explain positive adaptations observed in high-capacity states. This section moves beyond individual system descriptions to illustrate how the five systems interact under favorable conditions, producing emergent behavioral modes that cannot be attributed to any single system alone.
When energetic, neuroendocrine, and social conditions are favorable, the DBM predicts a shift toward exploratory, creative, and resilient behavioral modes. In such states, adequate EMS capacity supports sustained prefrontal engagement and cognitive flexibility; balanced RS activity permits rapid recovery following challenge; a stable Status System enables risk-taking without excessive threat sensitivity; a flexible Attention System allows fluid switching between internally generative (DMN) and externally focused (CEN) modes; and a well-functioning Connection System amplifies reward and motivation through social co-regulation.
Several high-capacity states deserve explicit attention:
- Flow emerges when EMS capacity is sufficient, AS salience is precisely tuned to the task, SS progress signals are clear, and CS safety is intact. The behavioral signature—effortless focus, high productivity, reduced self-consciousness—reflects not a special psychological state but a system configuration in which evaluation and energy provisioning are aligned with demand.
- Creativity and exploration occur when novelty signals are permitted, threat and recovery demands are low, and energy is available for experimentation. Creativity is, in DBM terms, a luxury behavior enabled by surplus capacity—one of the first functions shed when systems are constrained.
- Resilience reflects rapid RS recovery, flexible AS switching, and stable SS and CS signals. Resilient individuals recover faster not because they are “stronger” in some dispositional sense, but because their systems re-balance efficiently.
- Sustainable motivation occurs when wanting aligns with feasibility, energy provisioning is reliable, and outcome validation reinforces adaptive strategies. Motivation is not forced; it emerges naturally from system alignment.
From this perspective, creativity, resilience, and adaptive leadership are not personality traits alone but emergent properties of systems operating with sufficient spare capacity and low allostatic burden. The DBM thus provides a unified framework for understanding both collapse under strain and flourishing under supportive conditions.
4. Coupled Dynamics and Cascading Failure
The five systems rarely fail in isolation. Instead, they form coupled feedback loops that can drive the organism into self-reinforcing states of dysregulation.
4.1 The Neuroinflammatory Loop (RS → SS/CS)
Chronic RS activation → glucocorticoid resistance → systemic inflammation → cytokine effects on basal ganglia → dopamine suppression. This cascade:
- Weakens the SS (reducing sense of agency, rank, and capacity),
- Dampens CS-mediated social reward (making connection feel less rewarding),
- Promotes social withdrawal and passivity.
Withdrawal, in turn, increases isolation and perceived social threat, further activating the RS. This is a reinforcing loop that can underlie burnout, “atypical” depression, and certain anxiety presentations.
4.2 The Bioenergetic–Cognitive Loop (EMS → AS)
EMS depletion compromises inhibitory interneurons and PFC-dependent control. The AS becomes:
- More sensitive to noise,
- Less efficient at filtering distractions,
- Locked into high-alert salience states.
The resulting cognitive inefficiency increases metabolic demand, further depleting the EMS. Many experiences labeled “attention deficit,” “poor focus,” or “overwhelm” can be understood as this loop in action.
4.3 The Social Defeat Loop (SS → RS)
Perceived low status or repeated humiliation activates the HPA axis, increasing cortisol and sympathetic drive. Over time:
- Hippocampal and medial PFC remodeling reduces contextual judgment and flexibility,
- Amygdalar sensitization heightens threat detection, especially for social cues,
- The organism becomes vigilant for status threats and prone to defensive reactions.
This state feeds back into further social defeat experiences (e.g., perceived rejection, conflict, withdrawal), reinforcing the SS alarm and RS activation.
4.4 Additional Cross-System Interactions
Beyond these three canonical loops, the DBM anticipates multiple other interactions, such as:
- EMS-CS coupling (energy availability shaping capacity to engage socially),
- AS-SS coupling (attentional biases toward rank-relevant cues),
- RS-AS coupling (stress-driven shifts in network dynamics).
In practice, different individuals and contexts may be dominated by different loop configurations, explaining the heterogeneity of clinical presentations.
5. Clinical and Applied Implications: From Modification to Accommodation
The DBM suggests a shift in clinical logic: from forcing behavior change to restoring system capacity and renegotiating demands.
5.1 Assessment: Mapping the Five Systems
A DBM-informed assessment would:
- Screen for EMS constraints: chronic fatigue, sleep disruption, metabolic disease, medication effects.
- Map RS status: history of chronic stress, trauma, inflammatory conditions, autonomic symptoms.
- Evaluate SS dynamics: experiences of shame, social defeat, rank instability, workplace or family hierarchy.
- Characterize AS patterns: rumination, hypervigilance, distractibility, intolerance of ambiguity.
- Probe CS integrity: attachment history, perceived social safety, availability of mutually supportive relationships.
This approach reframes many “compliance” or “motivation” problems as mismatches between therapeutic demands and current system capacity.
5.2 Wanting Without Energy: A Central Clinical Phenomenon
A recurring pattern across clinical presentations is the state of wanting without energy: preparatory signals (anticipation, craving, goal-directed urges) are active, but the EMS denies energy provisioning due to feasibility constraints. The organism wants to act but cannot afford to.
This mismatch produces characteristic subjective states:
- Craving: Wanting signals persist without the capacity to act on them adaptively, driving escalation or substitution.
- Rumination: Preparatory processing continues without resolution, consuming metabolic resources in a high-cost, low-yield loop.
- Burnout frustration: The individual recognizes what needs to be done and feels urgency, yet experiences an inability to mobilize.
- Motivational paralysis: Wanting is intact, but the gap between signal and capacity produces helplessness and self-blame.
Clinically, these states are frequently misread as ambivalence, resistance, or secondary gain. The DBM interpretation is that wanting is a preparatory signal, not an authorization for action. When feasibility is insufficient, wanting without energy is the expected outcome—not a paradox requiring psychological explanation.
This reframing has direct treatment implications: rather than interpreting the wanting-doing gap as a target for motivational intervention, the clinician focuses on restoring the energetic and regulatory conditions under which wanting can be converted into action.
5.3 Bioenergetic Pacing and Demand Management
Interventions guided by DBM begin with bioenergetic pacing:
- Reducing unnecessary cognitive and emotional load,
- Improving sleep and circadian stability,
- Addressing nutritional deficits or medical contributors to mitochondrial exhaustion,
- Sequencing therapeutic tasks so that high-cost demands come after partial restoration of EMS capacity.
Rather than asking “How can we push this person harder?”, the DBM asks “What can we remove or simplify so that the system has enough spare capacity to adapt?”
5.4 Status Safety and Relational Design
For individuals with SS dysregulation, traditional hierarchies and authoritarian relational styles can be processed as survival threats. DBM-informed practice emphasizes:
- Reducing unnecessary status asymmetry in therapeutic and organizational contexts,
- Providing frequent signals of competence, contribution, and belonging,
- Avoiding shaming or comparative language that triggers social defeat circuits.
“Status safety” is not flattery; it is the deliberate design of interactions that minimize the neuroendocrine cost of being in a subordinate or scrutinized position.
5.5 Connection as a Primary Intervention
Given the CS mechanisms, interventions that increase safe, reciprocal connection are understood as biological leverage points, not soft, optional add-ons. This includes:
- Group-based interventions that foster mutual aid and shared identity,
- Family and team interventions that explicitly target trust and reliability,
- Practices that increase perceived proximity and co-regulation (e.g., regular check-ins, shared rituals).
By engaging oxytocin–dopamine heteromers, such interventions may help restore dopaminergic tone, indirectly supporting EMS, SS, and AS function.
5.6 Beyond the Clinic: Education, Work, and Policy
The DBM has implications for:
- Education: Recognizing that attention and motivation failures often reflect EMS and AS states, not moral failings.
- Workplace design: Structuring workloads, feedback, and hierarchies to minimize chronic RS activation and SS threat.
- Public health: Framing social isolation, precarious employment, and chronic stressors as biological risk factors for neurobehavioral dysfunction, not mere background variables.
6. Discussion
6.1 Positioning DBM Among Existing Models
The DBM overlaps with, but also extends, several major frameworks:
- Allostatic Load Models: DBM incorporates allostatic concepts within EMS and RS, but adds SS, AS, and CS to explain how stress is filtered, interpreted, and socially embedded.
- Polyvagal Theory: DBM embeds Polyvagal ideas in RS and CS but connects them to mitochondrial and network-level constraints, while acknowledging ongoing debates about its phylogenetic and mechanistic claims.
- Predictive Processing / Active Inference: DBM is compatible with prediction error minimization but emphasizes bioenergetic feasibility as a constraint on predictive updating and exploration. The Free Energy Principle (FEP) (Friston, 2010) deserves particular attention here, as its central construct—“free energy”—invites superficial comparison with DBM’s Energy Management System. However, the two are operating at different levels of description. FEP’s “free energy” is an information-theoretic quantity measuring prediction error; minimizing it means reducing surprise through perceptual updating or action. DBM’s EMS concerns literal bioenergetic capacity: mitochondrial function, ATP availability, and the metabolic cost of neural computation. The two frameworks may be complementary rather than competing: an organism may be minimizing prediction error (as FEP describes), but the EMS determines whether it has the metabolic resources to execute the actions that would accomplish that minimization. DBM can thus be read as specifying a bioenergetic constraint layer that FEP’s mathematical formalism abstracts away.
- Reinforcement Learning Models: DBM integrates dopaminergic RL with status and connection biology, explaining how social rank and affiliation shape reward sensitivity and learning.
The distinctive contribution of DBM is its explicit systems architecture, tying together energy, stress, status, attention, and connection as coupled, feedback-driven regulators of behavior.
6.2 Testable Predictions and Research Directions
The DBM generates multiple empirically testable hypotheses, such as:
- Multi-system signatures of burnout: Convergent evidence of mitochondrial impairment (EMS), glucocorticoid resistance (RS), striatal dopamine suppression (SS/CS), and salience network rigidity (AS) in burnout populations.
- Status interventions and energy allocation: Interventions that increase perceived status or contribution should produce measurable changes in bioenergetic markers and exploratory behavior.
- Connection interventions and dopaminergic tone: Enhanced social connection should increase indices of dopaminergic function and salience network flexibility, particularly in individuals with high inflammation or chronic stress.
- Network-level effects of energy restoration: Improving sleep and metabolic health should normalize SN–DMN–CEN switching and reduce symptoms currently labeled as “attention disorders.”
These predictions invite multi-modal research combining neuroimaging, endocrine and inflammatory markers, behavioral tasks, and ecological data.
6.3 Anticipated Misinterpretations
Because DBM challenges several deeply held assumptions in behavioral science, certain misreadings are predictable and worth addressing directly.
“DBM is biological determinism.” DBM does not deny agency; it explains the availability of agency. Choice exists within biological capacity. When capacity changes, the range of possible choices changes. DBM explains why agency fluctuates across states, not that it is absent.
“DBM excuses failure or removes responsibility.” Explaining constraints does not excuse harm or eliminate responsibility. DBM replaces ineffective moral pressure with actionable leverage: restoring capacity. Responsibility without capacity produces shame, not change; restoring capacity improves outcomes.
“DBM just renames reward.” DBM explicitly rejects a reward system. Reward signals exist, but they are informational, post-decisional, and non-causal. They do not initiate action, override feasibility, or function as goals. This is an architectural commitment, not a semantic reframing of reinforcement learning.
“DBM reduces everything to energy.” Energy is necessary but not sufficient. Behavior emerges from energy feasibility evaluated alongside threat, recovery, social value, attention, and connection. Energy enables action; it does not determine strategy.
“DBM is unfalsifiable or post-hoc.” DBM generates testable predictions through signal perturbation: altering neurotransmitters, hormones, sleep, nutrition, inflammation, or stress should systematically bias behavior in predicted directions, even when beliefs and context remain unchanged. Section 6.2 specifies several such predictions.
6.4 Limitations and Scope
The DBM is a conceptual framework, not yet a fully parameterized quantitative model. Limitations include:
- Incomplete mapping of all relevant molecular pathways,
- Potential oversimplification in reducing regulation to five systems,
- Limited current data integrating all five domains in the same cohort.
The model is intended as a scaffold for research and practice, to be refined as empirical findings accumulate.
Cultural and Contextual Variability. Although the DBM is grounded in evolutionarily conserved regulatory systems, the inputs and expressions of these systems are shaped by cultural context. For example, status signals may be derived from dominance, competence, age, spiritual authority, or relational harmony depending on sociocultural norms, while connection may prioritize autonomy, interdependence, or collective identity across societies.
The DBM treats Status and Connection as biological evaluative systems, not as fixed social scripts. Cultural environments modulate which cues are interpreted as salient, threatening, or rewarding, thereby shaping how these systems influence behavior. This flexibility allows the DBM to generalize beyond Western individualist contexts while remaining anchored in shared neurobiological mechanisms.
7. Conclusion
The Didomi Behavioral Model offers a systems biology framework for understanding behavior as an emergent property of five evolutionarily conserved regulatory systems: Energy Management, Recovery, Status, Attention, and Connection. By making explicit the feedback loops that couple these systems, the DBM reframes many “behavioral problems” as biological solutions to unsustainable internal equations.
Rather than viewing individuals as resistant, unmotivated, or irrational, the DBM invites clinicians, researchers, and policymakers to ask:
Given this organism’s current energy, stress, status, attention, and connection state, is this behavior actually the most adaptive solution available?
This shift, from moral or purely psychological interpretations to constraint-aware, biologically grounded understanding, opens new avenues for assessment, intervention, and prevention. Ultimately, the DBM seeks to bridge the gap between systems biology and everyday human behavior, offering a rigorous yet practical map of how bodies and brains negotiate the demands of being alive together.
Note on Nuance and Ongoing Debates. Several biological relationships described here, such as serotonergic modulation of social behavior and the hierarchical organization of autonomic responses, are active areas of research with ongoing debate. We present them as functional organizing principles within DBM, rather than as settled mechanistic endpoints, and expect their details to be refined as the empirical literature evolves.
Conflict of Interest Statement — The author declares no competing interests.
Funding — This work was conducted independently through Didomi Research with no external funding.
8. References
Beckes, L., & Coan, J. A. (2011). Social baseline theory: The role of social proximity in emotion and economy of action. Social and Personality Psychology Compass, 5(12), 976–988.
Cohen, S., Janicki-Deverts, D., Doyle, W. J., Miller, G. E., Frank, E., Rabin, B. S., & Turner, R. B. (2012). Chronic stress, glucocorticoid receptor resistance, inflammation, and disease risk. Proceedings of the National Academy of Sciences, 109(16), 5995–5999.
Devine, E. (2019). The neurobehavioral model. Eileen Devine.
Fanibunda, S. E., Deb, S., Maniyadath, B., & Vaidya, V. A. (2019). Serotonin regulates mitochondrial biogenesis via 5-HT2A receptor and SIRT1 in cortical neurons. Proceedings of the National Academy of Sciences, 116(22), 11028–11037.
Felger, J. C., & Miller, A. H. (2012). Cytokine effects on the basal ganglia and dopamine function: The subcortical source of inflammatory malaise. Frontiers in Neuroendocrinology, 33(3), 315–327.
Friston, K. (2010). The free-energy principle: A unified brain theory? Nature Reviews Neuroscience, 11(2), 127–138.
Grossman, P., & Taylor, E. W. (2007). Toward understanding respiratory sinus arrhythmia: Relations to cardiac vagal tone, evolution and biobehavioral functions. Biological Psychology, 74(2), 263–285.
Grueschow, M., et al. (2024). Locus coeruleus-norepinephrine system and salience network interaction. PMC.
Guidolin, D., Tortorella, C., Cervetto, C., Marcoli, M., Maura, G., & Agnati, L. F. (2025). Interaction between oxytocin and dopamine signaling: Focus on the striatum. International Journal of Molecular Sciences, 26(17), 8711.
Herman, J. P., McKlveen, J. M., Ghosal, S., Kopp, B., Wulsin, A., Makinson, R., Scheimann, J., & Myers, B. (2016). Regulation of the hypothalamic-pituitary-adrenocortical stress response. Comprehensive Physiology, 6(2), 603–621.
Holly, E. N., & Miczek, K. A. (2016). Ventral striatal dopamine and social status. Psychopharmacology, 233(2), 161–171.
Hudson, A. N., Van Dongen, H. P. A., & Honn, K. A. (2024). Mechanisms of sleep deprivation affecting prefrontal cortex function. PMC.
INFORMS Simulation Society (2016). Main tools in system dynamics. In Proceedings of the 2016 Winter Simulation Conference.
Jalbrzikowski, M., et al. (2023). Salience network rigidity and psychopathology. Frontiers in Human Neuroscience.
Karin, O., Raz, M., Tendler, A., Bar, A., Korem Kohanim, Y., & Alon, U. (2020). Dynamical compensation in the HPA axis explains its homeostatic range and dysregulation. Molecular Systems Biology, 16(12), e9510.
Menon, V. (2011). Large-scale brain networks and psychopathology: A unifying triple network model. Trends in Cognitive Sciences, 15(10), 483–506.
Patel, D., et al. (2019). Chronic social defeat stress induces sustained synaptic structural changes in the prefrontal cortex and amygdala. Behavioral Brain Research, 365, 137–145.
Paus, T. (2020). Status, serotonin, dopamine, and social rank. Philosophical Transactions of the Royal Society B, 375(1803), 20190444.
Picard, M., & McEwen, B. S. (2018a). Psychological stress and mitochondria: A conceptual framework. Psychosomatic Medicine, 80(2), 126–140.
Picard, M., & McEwen, B. S. (2018b). Brain mitochondrial bioenergetics change with behavior. Journal of Experimental Biology, 221(8), jeb176917.
Picard, M., et al. (2015). Mitochondrial functions modulate neuroendocrine, metabolic, inflammatory, and transcriptional responses to psychological stress. Proceedings of the National Academy of Sciences, 112(48), E6614–E6623.
Picard, M., et al. (2025). Brain energy management in mitochondrial disease. bioRxiv.
Porges, S. W. (2024). Polyvagal theory and the social engagement system. PMC.
Vitale, E. M., & Smith, A. S. (2022). Neurobiology of loneliness, isolation, and loss: Integrating human and animal perspectives. Frontiers in Behavioral Neuroscience, 16, 846315.
